Including a test of three types of queen: Wild Colony, Webster Russian, and VSH Italian
In the Spring of 2017, I decided to attempt treatment-free beekeeping of colonies managed for honey production. To do so, I stopped giving miticide treatments to the colonies that I keep in one of my apiaries and I started keeping detailed records on the fates of these colonies. Now, three years later, I am making an initial report on my progress toward having colonies that grow large, make honey, and survive without being treated with miticides. In addition, I will report on a one-year study conducted in 2019-2020 in which I compared colonies headed by three types of queen (wild caught in New York, Webster Russians from Vermont, and VSH Italians from California) regarding their suitability for treatment-free beekeeping.
My three-year (so far) study of treatment-free beekeeping
I am conducting this study near the small city of Ithaca, in New York State. It sits 170 miles northwest of New York City, at the southern end of Cayuga Lake. The surrounding landscape consists of rolling, open farmland to the north, and rugged, wooded hills to the south. The apiary that I have devoted to this study is located five miles east of Ithaca, and is tucked in a small valley called Ellis Hollow, which is where I have lived for most of my life. Ellis Hollow is a horseshoe-shaped valley that runs east-west, and is approximately 1 mile wide and 2.5 miles long. It is defined by two steep-sided hills—Mount Pleasant and Snyder Hill—which rise 500 feet above the valley’s floor along its northern and southern sides and on its eastern end; its western end is open (see Figure 1). The steep slopes and rounded tops of these hills are covered with forests, but the valley bottom is a mixture of fields, more forests, and wetlands along Cascadilla Creek.
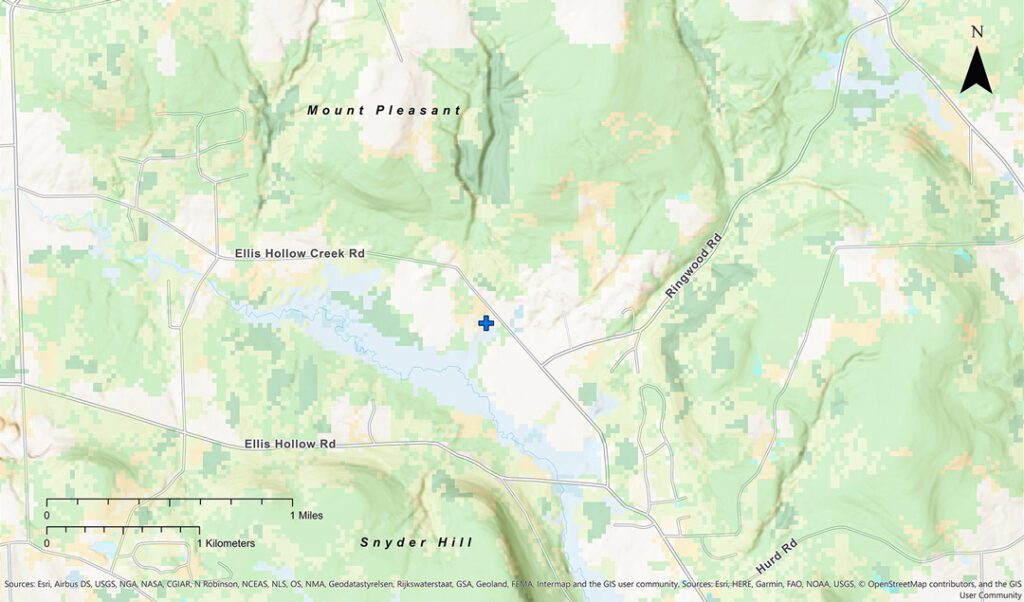
Ellis Hollow was settled in the early 1800s and until the 1940s it was home to about twenty small farms whose fields covered the valley’s arable land. These days, though, Ellis Hollow is mainly a rural residential community with some 150 homes, and most of the farms’ fields have grown back into woods and brushy places. Fortunately, many of the plants filling the abandoned fields—such as basswood (Tilia americana) and black locust (Robinia pseudoacacia) trees, pussy willow (Salix discolor) and staghorn sumac (Rhus typhina) shrubs, and patches of milkweed (Asclepias spp.) and goldenrod (Solidago spp.) plants—provide clean and plentiful forage for all sorts of insects, including honey bees.
There are approximately fifty colonies of honey bees living in Ellis Hollow. About twenty occupy beekeepers’ hives; the rest inhabit trees and buildings. Of the twenty or so colonies in hives, I keep about seventeen in my apiary, and two other beekeepers keep about four more. Regarding colonies in trees, I know that there are approximately 2.5 bee-tree colonies per square mile of forest around Ithaca (see chapter 2 of Seeley 2019), and I know that there are approximately ten square miles of forest on the hills that form the Ellis Hollow valley, so I estimate that there are about twenty-five wild colonies residing in hollow trees in this valley. As for wild colonies in the walls of buildings, I know of five sites—two in one neighbor’s house, and three in two other neighbors’ barns—where colonies have lived, off and on, for more than twenty years. I have no doubt that there are many more home sites of wild colonies in the houses and barns within this valley.
My apiary in Ellis Hollow occupies a clearing in the valley’s northeast corner (Figure 2). Forty years ago, this spot was the southeast corner of the pasture behind the house, barn, and sawmill of Omar Gleason, one of the old-timers who lived here from the 1910s to the 1970s. I knew Omar in the 1960s, when I was a boy. He and his family moved away in the 1970s and several years later their dilapidated buildings were burned down by our volunteer fire department. In 1986, I moved home to Ithaca to start work as a professor at Cornell. In setting up my laboratory, I needed to find several good apiary sites, so I was delighted to learn that the current owners of the Gleason place had just donated its acreage to the university. (Eventually, it became the Durland Bird Sanctuary, one of several natural areas in Ellis Hollow.) Wonderful! Soon I had permission to establish an apiary behind the stone foundation of the Gleason farmhouse, where the cast-iron pitcher pump still stands. I have kept colonies in this lovely spot for the past thirty-four years.

I keep my colonies in Langstroth hives, using deep hive bodies for both brood boxes and honey supers. Originally, I used 10-frame hives, but now I use mostly 8-frame equipment. The hives in my Ellis Hollow apiary are stocked with locally adapted bees. This is something that I have worked to achieve over the past five summers. My method has been to repopulate the hives in which colonies have died over winter with swarms caught in bait hives that I have set out each spring around the forested hills south of Ithaca (see Chapter 8 in Seeley 2017). I am confident that most of the swarms that move into my bait hives are from wild colonies living in these woodlands, because when my friends and I have gone bee hunting in the hills south of Ithaca, our beelines have always led us to wild colonies living in trees or buildings (e.g., a hunting cabin), (see, for example, the bee hunts described in Seeley 2017, and in Radcliffe and Seeley 2018).
How have I used the colonies in my apiary in Ellis Hollow? They have served primarily as producers of bees and brood for research projects and only incidentally as producers of honey. So my management of these colonies has been simple: in May, I give each colony one or two empty honey supers so it has room to store honey; and in early September, I remove any surplus honey from each colony, making sure that I leave each one with a top box that is stuffed with honey for winter stores. In most years, the colonies in this apiary will produce—despite the removals of bees and brood for various projects—about 1000 pounds of honey.
And how have I handled the threat of Varroa destructor to these colonies? Well, I should explain that these mites arrived in the Ithaca area in the mid 1990s. I first spied them on my bees in June 1994, and in August that year I saw a troubling sight in front of my hives: hundreds of workers crawling feebly through the grass, moving away from the hives. What I saw the following spring, in April 1995, was even more troubling: 89 percent of my colonies were dead, even though their hives were full of honey. This disaster spurred me to take action against Varroa, so in the summer of 1995 I began treating my colonies with fluvalinate (Apistan). When this miticide became ineffective a few years later, my students and I switched to treating our badly infested colonies (those with high mite counts in early August) with formic acid, oxalic acid, or a thymol-based medication.
In the spring of 2017, however, I decided to change course with the colonies in my Ellis Hollow apiary: no more miticide treatments. My students and I would use these colonies as before—as sources of bees and brood for our experiments, and of honey if they produced a surplus—but we would no longer treat them with miticides. We would, however, continue to make sure that each colony was well stocked with honey (i.e., its top hive body was full of honey) in mid-September, so that none would starve over winter. (Note: in the future, I may euthanize colonies that have high mite counts—more than 15 mites per 300 bees— in early September. This is because these colonies are apt to collapse in the autumn, and if this happens then their mites can be spread to the healthy colonies nearby through robbing. For more information on this “mite bomb” phenomenon, see Loftus et al. 2016, and Peck and Seeley 2018.)
Table 1 shows my records of the fates of the colonies in the Ellis Hollow apiary since 2017. The colony mortality over winter in this apiary has been about 30%, which is much higher than what I experienced here from the mid 1980s to the mid 1990s, i.e., before the arrival of Varroa. Back then, I had 10-15% colony mortality over winter at this site. (Note: there were, however, two exceptional winters—1990-91 and 1991-92—when I experienced ca. 80% colony mortality across all my apiaries. These were the winters that followed the arrival of the tracheal mite, Acarapis woodi, in the Ithaca area. Fortunately, this problem subsided quickly. I suspect that it did so thanks to strong natural selection for bees with resistance to tracheal mites.)
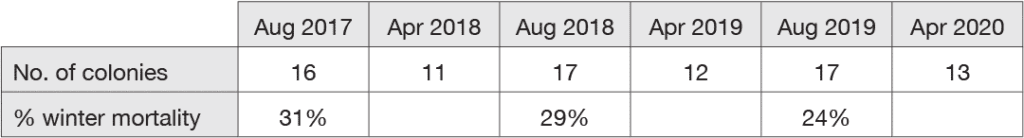
I have little doubt that the ca. 30% mortality of colonies shown in Table 1 is a result of high populations of Varroa (and ensuing damage from viruses) in some of the colonies. I say this because I have found that in these untreated colonies, the mite level in a colony in September is a very good predictor of whether this colony will be dead (or alive) the following April. For example, on 18 Sept 2018, I measured the mite loads (mites/300 bees, powdered sugar test) of the 17 colonies in the Ellis Hollow apiary, and on 25 April 2019, I inspected these colonies to see which had died. The results, shown in Table 2, show that there was great variation among the colonies in their mite loads in September 2018, and that this variation was tightly associated with which colonies were dead or alive in April 2019.

In summary, what I have seen so far in my apiary in Ellis Hollow—where I have ceased treating the colonies with miticides, and where each summer I have rebuilt my colony numbers using swarms caught in bait hives—is that 24-31% of the colonies have died over winter. I have also seen that the colonies that have died over winter are the ones with high mite counts in September. This 24-31% level of winter colony mortality is much higher than what I experienced in the 1970s and 1980s (two decades without Varroa), and it is certainly not ideal. Nevertheless, I will persist with this experiment. I am motivated to do so because I enjoy not dosing the Ellis Hollow colonies with miticides, and because I enjoy catching swarms that, as we shall see next, often produce colonies that are able to control the Varroa mites. So I remain optimistic that eventually most of the colonies in my Ellis Hollow apiary will possess a satisfactory ability to control Varroa.
My one-year test of three types of queens
As explained above, my program of treatment-free beekeeping is based on capturing swarms in bait hives I have placed in the forested hills south of Ithaca, that is, in locations far from beekeepers’ colonies. I have assumed that these swarms tend to produce colonies that can thrive without treatments to control Varroa destructor. In 2019, I decided to test this assumption by conducting an experiment. I set up twenty colonies that started out as closely matched as possible except with respect to their queens, which were of three types: Wild Caught by me in New York State; Russians produced by Kirk Webster, a successful commercial beekeeper in Vermont who does not treat for Varroa (see Webster 2015, and Rinderer and Coy 2020); and VSH Italians from a large queen producer in California who does treat for Varroa. In mid-June 2019, I received five Webster Russian queens and seven VSH Italian queens by mail. Already, I had eight Wild Caught queens that I had acquired when swarms occupied eight of the bait hives that I had set out (as described above) in mid-April 2019.

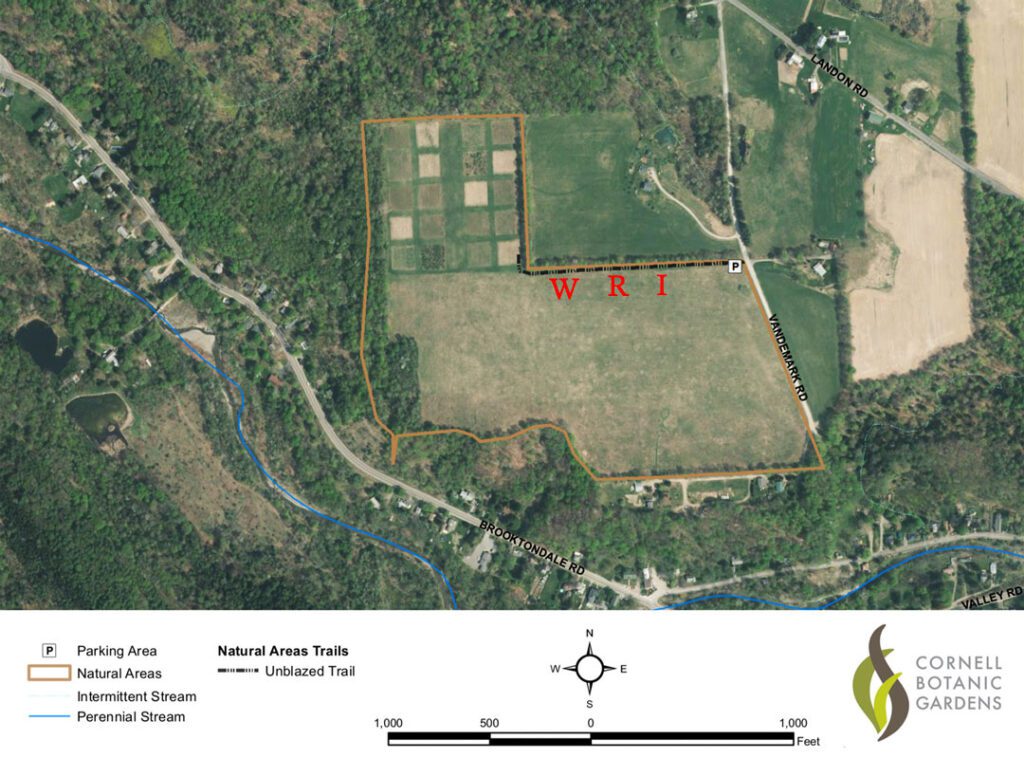
On 19-20 June, 2019, I introduced these twenty queens (using push-in cages; see Sammataro and Avitabile 2011) into twenty small, queenless colonies that were housed in five-frame hives. Each colony’s hive contained two frames of comb covered with bees and nearly filled with brood, one frame of comb nearly filled with pollen and honey, and two frames of empty comb. The forty frames of bees and brood that I used to establish these twenty test colonies came from ten source colonies living in the other two apiaries that I have besides the one in Ellis Hollow. Each source colony provided the bees and brood for two test colonies, and the two test colonies from each source colony were assigned to two different test groups (e.g. Wild Caught and Webster Russian, or Webster Russian and VSH Italian, etc.). I moved all twenty test colonies to a shared site where I arranged them in three clusters (one for each queen type). These clusters were separated by more than 100 feet (Fig. 3). The shared site was the Dunlop Meadow near Brooktondale, New York (see https://cornellbotanicgardens.org/wp-content/uploads/2018/10/Dunlop-Meadow.pdf), and the three clusters were set up along the 880- foot-long hedgerow that runs east-west along the meadow’s northern boundary (Fig. 4). Every colony’s hive faced south, and every colony’s hive color was unique within its cluster.
On 3 July 2019, I transferred all twenty colonies to ten-frame hives, and I put a dot of white paint on the thorax each colony’s queen. On 8 July (18 days after the colonies were established), I inspected each colony and measured its brood area (number of frame sides filled with brood). Here is what I found: colonies with a Wild Caught queen, 3.9±0.6 frame sides; colonies with a Webster Russian queen, 3.0±0.3 frame sides; and colonies with a VSH Italian queen: 3.5±0.5 frame sides. I also found that all twenty colonies were queenright. Seeing this, I decided to not disturb the colonies for the next several weeks.
On 27 August 2019, I inspected each colony, to see if it was still queenright (and if so, whether or not it contained its original queen) and was thriving, i.e., it had a good brood pattern and good honey stores. Every colony was queenright, and still had its original queens, except one colony in the VSH Italian group that had replaced its queen. In this colony, I found an unmarked queen laying eggs and an open queen cell.
On 9 October 2019, I inspected each colony again. In doing so, I checked (1) whether or not the colony was queenright and (if so) whether or not it had its original queen, (2) whether it was strong or weak (i.e., whether bees filled the hive or covered only some of the frames), and (3) its mite load (i.e., mites per 300 bees, measured using the powdered sugar method). Table 3 summarises what I found. We see that two of the eight colonies in the Wild Caught group were queenless and weak (with bees covering just two-four frames), but that all the other colonies were queenright and strong, with bees on all ten frames. We also see that among the queenright colonies, two out of six in the Wild Caught group, two out of five in the Webster Russian group, and one out of seven in the VSH Italian group had an unmarked queen. These were either supersedure queens or replacement queens reared in colonies that had swarmed in August or September. (In the Ithaca area, about 20% of swarming occurs in late August and early September [Fell et al. 1977]). What is most striking, however, are the differences that we see among the three groups in their mite counts. The mean (± SD) mite counts for the colonies with Wild Caught, Webster Russian, and VSH Italian queens are 4.6 ± 6.7, 1.6 ± 1.2, and 15.3 ± 6.0 mites per 300 bees. The difference between the mean counts for the Wild Caught (WC) and Webster Russian (WR) queen colonies is not significant (p > 0.05), but the differences between the mean counts for these two types of colonies and the mean count for the colonies with VSH Italian queens are both significant (WC vs. VSH Italian, p < 0.01; WR vs. VSH Italian p < 0.001).
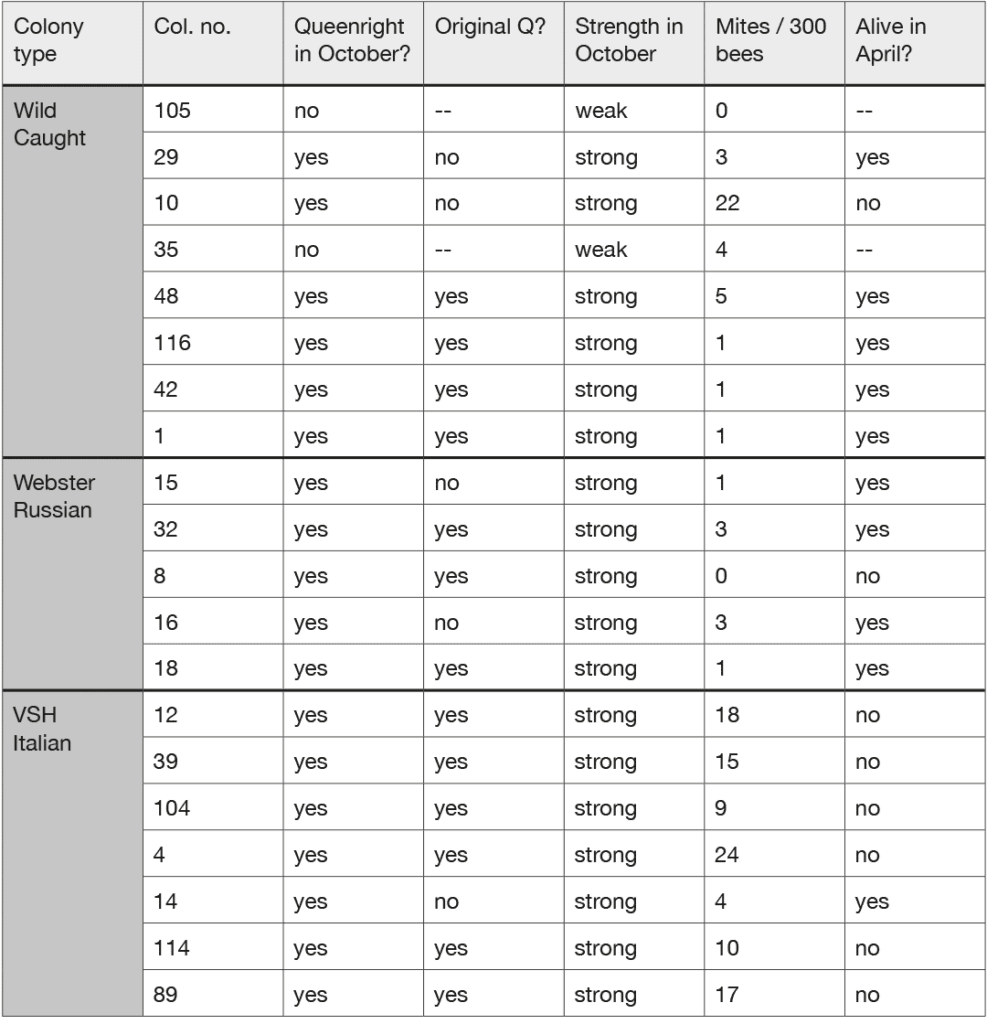
Seven months later, on a warm Spring day in Ithaca (7 April 2020), I again inspected all the colonies. Table 3 shows what I found: of the six Wild Caught colonies that went into winter queenright, all but one was alive; of the five Webster Russian colonies (all of which went into winter queenright), all but one was alive; and of the seven VSH Italian colonies (all of which went into winter queenright), only one was alive. These results are best summarized in terms of the percentages of the queenright colonies that survived winter: Wild Caught, five out of six colonies (83%); Webster Russian, four out of five colonies (80%), and VSH Italian, one out of seven colonies (14%).
We cannot draw sweeping conclusions from this study, for it involved only twenty colonies and it unfolded over just one year. Nevertheless, I think it is useful to summarise its key findings, for they are least suggestive of where a beekeeper who wishes to pursue treatment-free beekeeping should get his or her queens.
- There was greater heterogeneity—with respect to queen failures over summer and mite counts in October—among the colonies with Wild Caught queens than among those with Webster Russian and VSH Italian queens.
- On average, the mite counts in October were markedly lower in the colonies with Wild Caught queens and Webster Russian queens relative to the colonies with VSH Italian queens.
- Among the colonies that were queenright going into winter, the percentage that survived winter was markedly higher for the colonies with Wild Caught and Webster Russian queens than for the colonies with VSH Italian queens.
- Among the seven colonies with VSH Italian queens, there was just one (Colony 4) that had a low mite count in October and that survived winter; this was the colony that changed its queen in August or September.
I wonder, regarding point 4, did the one colony in the VSH Italian group that survived winter do so because the process of swarming (or supersedure) reduced this colony’s mite load (see Seeley and Smith 2016), or because its new queen acquired genes for mite-resistance when she mated with drones from wild colonies in the area, or both? I look forward to trying to test the second possibility. Does an abundance of wild colonies in the region where a queen conducts her mating flight(s) help to endow her colony with strong resistance to Varroa destructor, by enabling her to mate primarily with drones from colonies that are thriving without being treated for these mites? Stay tuned!
References
Fell, R.D., J.T. Ambrose, D.M. Burgett, D. De Jong, R.A. Morse, and T.D. Seeley. 1977. The seasonal cycle of swarming in honeybees. Journal of Apicultural Research 16: 170-173.
Loftus, C.L., M.L. Smith, and T.D. Seeley. 2016. How honey bee colonies survive in the wild: testing the importance of small nests and frequent swarming. PLoS ONE 11(3): e0150362. doi:10.1371/journal.pone.0150362.
Peck, D.T. and T.D. Seeley. 2019. Mite bombs or robber lures? The roles of drifting and robbing in Varroa destructor transmission from collapsing honey bee colonies to their neighbors. PLoS ONE 14(6): e0218392. https://doi.org/10.1371/journal.pone.0218392
Radcliffe, R.R. and T.D. Seeley. 2018. Deep forest bee hunting: a novel method for finding wild colonies of honey bees in old-growth forests. American Bee Journal 158(August): 871-877.
Rinderer, T.E. and S.E. Coy. 2020. Russian Honey Bees. Salmon Bayou Press.
Sammataro, D. and A. Avitabile. 2011. The Beekeepers Handbook. 4th Edition. Cornell University Press, Ithaca, New York.
Seeley. T.D. 2017. Following the Wild Bees. Princeton University Press, Princeton, New Jersey.
Seeley, T. D. 2019. The Lives of Bees. Princeton University Press, Princeton, New Jersey.
Seeley, T.D. and M.L. Smith. 2015. Crowding honeybee colonies in apiaries increases their vulnerability to the deadly ectoparasitic mite Varroa destructor. Apidologie 46:716-727.
Webster, K. 2015a. Commercial beekeeping without treatments of any kind–putting the pieces together. Part I of Two Parts. American Bee Journal 145(March): 203-206.
Webster, K. 2015b. Commercial beekeeping without treatments of any kind–putting the pieces together. Part II of Two Parts. American Bee Journal 145(April): 312-315.